Invasion History
First Non-native North American Tidal Record: 2018First Non-native West Coast Tidal Record:
First Non-native East/Gulf Coast Tidal Record: 2018
General Invasion History:
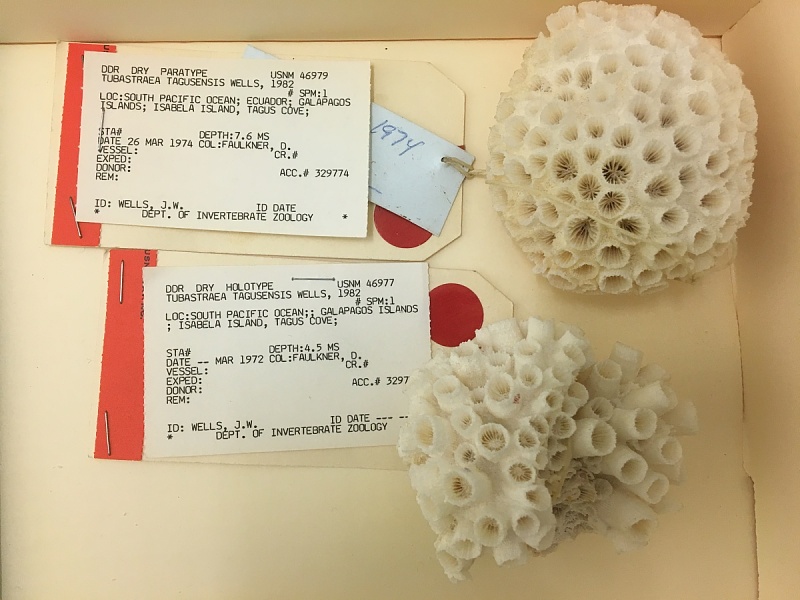
Tubastraea tagusensis (Tagus Cove Cup Coral) was described from Tagus Cove on Isabela Island, in the Galapagos Archipelago. Elsewhere in the tropical Eastern Pacific it has been collected on Socorro Island, in the Revillagigedo Archipelago, off Mexico (Wells 1982; Creed et al. 2023; US National Museum of Natural History Collections 2023). This coral is abundant in the Galapagos, but records in the Indo-West Pacific are scattered, from Hong Kong and Palau, to the Persian Gulf. Creed et al. (2017) considered the Indo-West Pacific record to be cryptogenic, and possibly misidentifications. In 2000, T. tagusensis was collected off Ilha Grande, in Rio de Janeiro State, Brazil, and spread rapidly along the coast of Brazil, especially occurring on shipwrecks and oil platforms, but also colonizing rocky shores and outcrops. It has spread south to Sao Paulo (~25° S) to Ceara State (2° S) (Figueira de Paula and Creed 2004; Creed et al. 2017). Before 2018, Tubastraea tagusensis was collected at several oil-drilling areas in the Gulf of Mexico, Texas in 2016–2018 (Figueroa et al. 2019; Soared et al. 2020). The extent of T. tagusensis' range in the Gulf of Mexico is not known.
North American Invasion History:
Invasion History on the Gulf Coast:
Tubastraea tagusensis (Tagus Cove Cup Coral) was collected by diving in 2017–2018 from 3 former oil-drilling sites, now maintained as artificial reefs, at 20–49 m depth. Specimens of Tubastraea spp. were identified by morphological and genetic methods; 95 specimens of T. coccinea, 71 specimens of T. tagusensis, and 3 of an indeterminate clade (Figuroa et al. 2019). However, the spatial extent and abundance of Tubastraea tagusensis in the Gulf of Mexico is not known.
Invasion History Elsewhere in the World:
Tubastraea tagusensis (Tagus Cove Cup Coral) is an azooxanthellate (lacks symbiotic algae) described from Tagus Cove, on Isla Isabela, in the Galapagos Islands. It is widespread an abundant in the Galapagos. It has also been collected on Isla Socorro, off the Pacific coast of Mexico. Other Indo-Pacific records, off Palau, in the Nicobar Islands, India, Hong Kong, and in the Persian Gulf, have been considered cryptogenic, because of the diversity of the genus in the Indo-West Pacific, and may be misidentifications (Creed et al. 2017). Genetic studies of this genus in the Pacific are desirable.
In 2000, T. tagusensis was collected off Ilha Grande, in Rio de Janeiro state, Brazil. On the Brazilian coast, this coral now occurs from Sao Paulo State (25°S) to Ceara State (2°S). often on shipwrecks and oil platforms, but also on rocky shores (Soares et al. 2020). In Brazil, it has been often found to replace or overgrow native corals and sponges (Creed et al. 2017). In 2016–2018, T. tagusensis was collected at several oil exploration sites in the Gulf of Mexico, off Texas (Figueroa et al. 2019). The extent of its range in Gulf of Mexico waters is not known.
Description
Tubastraea tagusensis (Tagus Cove Cup Coral) is an azooxanthellate coral (lacking symbiotic algae), which grows in colonies consisting of clumps of calcareous cups, projecting from a spongy calcareous base. The corallite (calcareous wall around a single polyp) is cylindrical, up to 8–10 mm in diameter, and may rise 3–15 mm above the colony skeleton (coenosteum). The corallites form convex, globular colonies 70–100 mm in diameter and 70–100 mm tall. The corallite walls are thin, with low, thin, smooth or slightly grainy ribs. The interior of the corallite is partly divided by septa, projecting into the body cavity. There are two kinds of septa of varying width, alternating in the calyx (cup) of the corallite, which are arranged in three cycles around the opening of the corallite. The septa do not protrude on the exterior of the corallite. The columella—a projection of the skeleton in the center of the corallite—varies with size, from a weak tangle of septa to a prominent structure filling half the diameter of the calyx. The polyps vary in color, from lemon-yellow, with red around the mouth, or pale red-violet, with yellow tissue overgrowing the skeleton (Wells 1982).
Taxonomy
Taxonomic Tree
| Kingdom: | Animalia | |
| Phylum: | Cnidaria | |
| Class: | Anthozoa | |
| Subclass: | Hexacorallia | |
| Order: | Scleractinia | |
| Suborder: | Dendrophylliina | |
| Family: | Dendrophylliidae | |
| Genus: | Tubastraea | |
| Species: | tagusensis |
Synonyms
Potentially Misidentified Species
Tubastraea coccinea (Orange Cup or Orange Sun Coral) is widely distributed in the tropical Pacific, and was introduced to the Gulf of Mexico by 1950. It is now established from Sao Paulo state, Brazil to the Gulf of Mexico (Creed et al. 2017).
Tubastraea micranthus
Tubastraea micranthus has a broad distribution in the tropical Indo-West Pacific. In 2006, it was found growing on oil platforms off Louisiana, in the Gulf of Mexico (Creed et al. 2017).
Ecology
General:
In the Galapagos Archipelago, Tubastraea tagusensis is abundant on rock outcrops and overhangs at 3–43 m depth (Wells et al. 1982). Tubastraea spp. lack zooxanthellae (symbiotic algae), and so are not dependent on photosynthesis. Because they are not limited by light, they can occur at depths down to 60m (Shearer 2011). They feed on zooplankton captured with their tentacles (Barnes 1983). Planulae of Tubastraea tagusensis in Brazil settled equally on plates made with wood, granite, concrete, steel, and ceramic tiles, with a slight preference for cement over steel (Creed and Figueira de Paula 2007). On the coast of Brazil, T. tagusensis is often most abundant on oil platforms and other structures, but is also abundant on coral reefs and rock outcrops, up to the lower intertidal (Silva et al. 2019). Most occurrences in the Western Atlantic have been on abandoned on oil platforms (Cairns 2000; Fenner and Banks 2004; Creed and Figueira de Paula 2007). In experiments, T. tagusensis survived exposure to temperatures as low as 15 °C for 96 hours, but died in 24-hour exposure at 12.5 °C. Its distribution on Cabo de Frio Island, Brazil, was strongly affected by an adjacent cool-water upwelling zone, and was rare or absent where temperatures dropped below 15 °C (Batista et al. 2016). We haven't found definite numbers on its upper temperature tolerance, but populations in the Galapagos apparently disappeared after an El Niño event, but reappeared 20 years later (Glynn and De Weerdt 1991, cited by Creed et al. 2017). Tubastraea tagusensis survived transfers to 15 PSU without mortality for 30 days, (Moreira et al. 2014), although we have not found records from strongly brackish waters.
Food:
Zooplankton, epibenthos
Consumers:
nudibranchs
Competitors:
Tubastraea coccinea, other corals
Trophic Status:
Suspension feederHabitats
| General Habitat | Coral reef | None |
| General Habitat | Bedrock | None |
| General Habitat | Rocky | None |
| General Habitat | Marinas & Docks | None |
| Salinity Range | Polyhaline | 18-30 PSU |
| Salinity Range | Euhaline | 30-40 PSU |
| Tidal Range | Subtidal | None |
| Vertical Habitat | Epibenthic | None |
Life History
Tubastraea tagusensis (Tagus Cove Cup Coral) is an azooxanthellate coral (lacking symbiotic algae), which grows in colonies consisting of clumps of calcareous cups, projecting from a spongy calcareous base, forming convex, globular colonies 70–100 mm in diameter and 70–100 mm tall. Polyps reproduce asexually by budding from the base or from the oral disk to produce new polyps, which secrete their own calcareous skeletons (Barnes 1983; Harrison 2011). Fragments of damaged polyps can grow into whole polps, from small pieces of tissue (Luz et al. 2018). Polyps of this coral are hermaphroditic, and release sperm into the water, but brood their eggs, which develop into planula larvae. This coral can also produce planulae asexually (Creed et al.2017). Tubastraea tagusensis, in Brazil, had more frequent and more closely spaced budding, and somewhat irregular colonies (de Paula and Creed 2004). The planulae are brooded, and quite large (1–3 mm long) and can be visible during mass-spawning events. They can remain competent to settle for up to 18 days, so have the capability for long-distance dispersal by currents (Paz-Garcia et al. 2007; de Paula and Creed 2014).
Tolerances and Life History Parameters
| Minimum Temperature (ºC) | 15 | Experimental. Battsta et al. 2017 |
| Minimum Salinity (‰) | 15 | Moreira et al. 2014 |
| Maximum Width (mm) | 70 | Colony width (Wells 1982) |
| Maximum Height (mm) | 70 | Colony height (Wells 1982) |
General Impacts
Ecological Impacts
Tubastraea tagusensis (Tagus Cove Cup Coral), like T. coccinea (Orange Cup or Orange Sun Coral), has been an aggressive colonizer on the coast of Brazil, replacing native corals and sponge on rocky reefs, together with and sometimes dominating T. coccinea, decreasing taxonomic and functional diversity on reefs (Lages et al. 2011; Pires-Teixeira et al. 2021). In field experiments, the native corals Siderastrea stellata and Montastraea cavernosa suffered tissue mortality when in contact with T. tagusensis (Miranda et al. 2016). Invasions by T. tagusensis also affect the abundance and diversity of mobile invertebrates (Silva et al. 2019). Coral invasions also affect the feeding rates of both herbivorous and invertebrate-feeding fishes (Miranda et al. 2019).
Economic impacts
In Brazil, invasions of Tubastraea spp. are perceived as negatively affecting the biodiversity, visual quality, and fisheries of rocky reefs and coral reefs of coastal waters (Dutra et al. 2023). Consequently, there have been efforts to monitor its distribution and control it through local eradication (Creed et al. 2017; Dutra et al. 2023). Methods have included the use of fresh or low-salinity water (Moreira et al. 2014), wrapping colonies (Mantelatto et al. 2015), or use of sodium hypochlorite (Altvater et al. 2017).
Regional Impacts
| SA-III | None | Ecological Impact | Competition | ||
In experiments in Todos de Santos Bay, abundance of Tubastraea tagusensis was associated with reduced abundance of native corals. In transplantation experimenrts, contact with T. tagusensis led to the mortality of adjacent native coral polyps (Miranda et al. 2016). |
|||||
| NWP-2 | None | Ecological Impact | Competition | ||
Tubastreaa tagusensis, often together with T. coccinea, is often associated with a reduced abundance of native corals in Ilha Grande, Brazil (Lages et al. 2011). |
|||||
| NWP-2 | None | Ecological Impact | Food/Prey | ||
Tubastraea tagusensis and T. coccinea both produce substances which reduce feeding by generalist fish species, and also inhibited the growth of native algae (Lages et al. 2010). |
|||||
| SA-III | None | Ecological Impact | Food/Prey | ||
In Todos dos Santos Bay, Dense cover of Tubastraea tagusensis and T. coccinea was associated with reduced feeding by territorial and free-moving reef fishes (Miranda et al. 2018). |
|||||
| SA-II | None | Ecological Impact | Competition | ||
On Ilha Grande, Brazil, increases in the cover of Tubastraea spp. was associated with a decrease in the diversity of the benthic community on rocky reefs, through competition for space (Lages et al. 2011). |
|||||
| SA-II | None | Ecological Impact | Food/Prey | ||
Tubastraea tagusensis and T. coccinea both produce substances which reduce feeding by generalist fish species, and also inhibited the growth of native algae (Lages et al. 2010). |
|||||
| SA-II | None | Ecological Impact | Habitat Change | ||
High densities of Tubastraea spp. on Brazilian rocky reefs are associated with a decrease in the abundance of mobile invertebrates, compared to diverse communtiies with macoralgae and native fouling invertebrates (Silva et al. 2019). |
|||||
| SA-II | None | Economic Impact | Ecosystem Services | ||
In Brazil, invasions of Tubastraea spp. are perceived as negatively affecting the biodiversity, visual quality, and fisheries of rocky reefs and coral reefs of coastal waters (Dutra et al. 2023). Consequently, there's been efforts to monitor its distribution and efforts at controlling it through local eradication (Creed et al. 2017; Dutra et al. 2023). Methods have included the use of fresh or low-salinity water (Moreira et al. 2014), wrapping colonies (Mantelatto et al. 2015), or use of sodium hypochlorite (Altvater et al. 2017). |
|||||
Regional Distribution Map
Non-native
Native
Cryptogenic
Failed
Occurrence Map
References
Soares, Marcelo de Oliveira Davis, Marcus Carneiro, Pedro Bastos de Macêdo (2018) Northward range expansion of the invasive coral (Tubastraea tagusensis) in the southwestern Atlantic, Marine Biodiversity 48: 1651–1654Altvater, Luciana de Messano, Luciana V. R. Andrade, Maurício Apolinário, Moacir Coutinho, Ricardo (2017) Use of sodium hypochlorite as a control method for the non-indigenous coral species Tubastraea coccinea Lesson, 1829, Management of Biological Invasions 8(2): 197-204
Barbosa, Andreia C. C. Vinagre, Catarina Mizrahi, Damián Flores, Augusto A. V. (2019) Temperature-driven secondary competence windows may increase the dispersal potential of invasive sun corals, Marine Biology 166: Published online
https://doi.org/10.1007/s00227-019-3580-7
Barnes, Robert D. (1983) Invertebrate Zoology, Saunders, Philadelphia. Pp. 883
Batista, Daniela and 9 authors (2017) Distribution of the invasive orange cup coral Tubastraea coccinea Lesson, 1829 in an upwelling area in the South Atlantic Ocean fifteen years after its first record, Aquatic Invasions 12(1): 23-32
DOI: https://doi.org/10.3391/ai.2017.12.1.03
Braga, Marcus Davis Andrade et al. (2021) Retirement risks: Invasive coral on old oil platform on the Brazilian equatorial continental shelf, Marine Pollution Bulletin 165(112156): Published online
https://doi.org/10.1016/j.marpolbul.2021.112156
Cairns, Stephen (2000) A revision of the shallow-water azooxanthellate scleractinia of the Western Atlantic, Studies on the fauna of Curacao and other Caribbean Islands. 125: 1-235
Capel, Kátia C. C.;Creed, Joel C ;Kitahara, Marcelo V. (2020) Invasive corals trigger seascape changes in the southwestern Atlantic, Bulletin of Marine Science 96(1): 217-218
https://doi.org/10.5343/bms.2019.0075
Costa, Thiago J.F. and 10 authors (2014) Expansion of an invasive coral species over Abrolhos Bank, Southwestern Atlantic, Marine Pollution Bulletin 85: 252-253
Creed, J. C. (2006) Two invasive alien azooxanthellate corals, Tubastraea coccinea and Tubastraea tagusensis, dominate the native zooxanthellate Mussismilia hispida in Brazil, Coral Reefs 25: 350
Creed, Joel C. and 11 authors (2017) The invasion of the azooxanthellate coral Tubastraea (Scleractinia: Dendrophylliidae) throughout the world: history, pathways and vector, Biological Invasions 19: 283-395
DOI 10.1007/s10530-016-1279-y
Creed, Joel C.; Figueira de Paula, Alline (2007) Substratum preference during recruitment of two invasive alien corals onto shallow-subtidal tropical rocky shores., Marine Ecology Progress Series 330: 101-111
da Silva, Amanda Guilherme; Figueira de Paula, Alline; Fleury, Beatriz Grosso; Creed, Joel Christopher (2014) Eleven years of range expansion of two invasive corals (Tubastraea coccinea and Tubastraea tagusensis) through the southwest Atlantic (Brazil), Estuarine, Coastal and Shelf Science 141: 9-16
de Oliveira Soares, Marcelo Salani, Sula Paiva, Sandra Vieira Andrade Braga, Marcus Davis (2020) Shipwrecks help invasive coral to expand range in the Atlantic Ocean, Marine Pollution Bulletin 158(111394): Published online
https://doi.org/10.1016/j.marpolbul.2020.11139
Dutra, Beatriz Sant’Anna Vasconcelos Marafiga et al. (2023) When species become invasive research becomes problem oriented: a synthesis of knowledge of the stony coral Tubastraea, Biological Invasions 25: 2069 - 2088
https://doi.org/10.1007/s10530-023-03032-4
Fenner, Douglas; Banks, Kenneth (2004) Orange cup coral Tubastraea coccinea invades Florida and the Flower Garden banks, Northwestern Gulf of Mexico., Coral Reefs 23: 505-507
Figueira de Paula, Alline; de Oliveira Pires, Debora; Creed, Joel Christopher (2014) Reproductive strategies of two invasive sun corals (Tubastraea spp.) in the southwestern Atlantic, Journal of the Marine Biological Association of the United Kingdom 94(3): 481-492
Figueira de Paula; Creed, Joel Christopher (2004) Two species of the coral Tubastraea (Cnidaria, Scleractinia) in Brazil: a case of accidental introduction, Bulletin of Marine Science 74: 175-183
Figueroa, Diego F.; McClure, Amelia; Figueroa, Nicole J.; Hicks, David W. (2019) Hiding in plain sight: invasive coral Tubastraea tagusensis (Scleractinia:Hexacorallia) in the Gulf of Mexico, Coral Reefs 38: 395–403
https://doi.org/10.1007/s00338-019-01807-7
Guilhem, Isabella F. Masi, Bruno P. Creed, Joel C. (2020) Impact of invasive Tubastraea spp. (Cnidaria: Anthozoa) on the growth of the space dominating tropical rocky-shore zoantharian Palythoa caribaeorum (Duchassaing and Michelotti, 1860), Aquatic Invasions 18(1): 98-113
https://doi.org/10.3391/ai.2020.15.1.07
Harrison, Peter L. (2011) Sexual reproduction of scleractinian corals, In: Dubinsky, Z.; Stambler, N. (Eds.) Coral Reefs: An Ecosystem in Transition. , <missing place>. Pp. 59-185
DOI 10.1007/978-94-007-0114-4_6,
Hoeksema; Bert W.; Samimi-Namin, Kaveh McFadden, Catherine S. ; Rocha, Rosana M.; van Ofwegen, Leen P. ; Hiemstra,, Auke-Florian; Vermeij. Mark J. A. (2023) Non-native coral species dominate the fouling community on a semi-submersible platform in the southern Caribbean, Marine Pollution Bulletin 194(115353): Published online
Lages, Bruno G.; Fleury, Beatriz G.; Pinto, Angelo C.; Creed, Joel C. (2010) Chemical defenses against generalist fish predators and fouling organisms in two invasive ahermatypic corals in the genus Tubastraea, Marine Ecology 31: 473-482
Luz, Bruna L. P.; Di Domenico, Maikon; Migotto, Alvaro E.; Kitahara, Marcelo V. (2019) Life-history traits of Tubastraea coccinea: Reproduction, development, and larval competence, Ecology and Evolution 10: 6223–6238
DOI: 10.1002/ece3.6346
Mantelatto, M. C.; Creed, J. C. ; Mourao, G. G.; Migotto, A. E.; Lindner, A. (2011) Range expansion of the invasive corals Tubastraea coccinea and Tubastraea tagusensis in the Southwest Atlantic, Coral Reefs 30: 397
Mantelatto, Marcelo C.; Creed, Joel C. (2015) Non-indigenous sun corals invade mussel beds in Brazil, Marine Biodiversity 45: 605-606
Merz, Ewa; Kitahara, Marcelo V. ; Flores, Augusto A.V. (2023) A legacy of invasive sun corals: Distinct mobile invertebrate assemblages at near-reef coral-dominated rubble, Marine Environmental Research 1888(105974): Published online
https://doi.org/10.1016/j.marenvres.2023.105974
Meurer, Bruno C.; Lages, Nicolas S.; Pereira, Oliver A.; Palhano, Samantha Magalhaes, Gabriela M. (2010) First record of native species of sponge overgrowing invasive corals Tubastraea coccinea and Tubastraea tagusensis in Brazil, Marine Biodiversity Records 3: published online
Miranda, Ricardo J. Cruz, Igor C. S. Barros, Francisco (2016) Effects of the alien coral Tubastraea tagusensis on native coral assemblages in a southwestern Atlantic coral reef, Marine Biology 163: Published online
Miranda, Ricardo J. de Anchieta. José Nunes, C. C. Mariano-Neto, Eduardo Sippo, James Z. Barros, Francisco (2018) Do invasive corals alter coral reef processes? An empirical approach evaluating reef fish trophic interactions, None <missing volume>: <missing location>
Mizrahi, Damián; Silva, Milena C.; Fonseca, Maurício L.; Lopes, Rubens M. (2023) Resistance to desiccation and healing regeneration in the sun coral, Bioinvasions Records 14: In press
Moreira, Patricia L. Ribeira, Felipe Creed, Joel C. (2014) Control of invasive marine invertebrates:an experimental evaluation of the use of low salinity for managing pest corals (Tubastraea spp.), Biofouling 5(5): 639-650
https://doi.org/10.1080/08927014.2014.906583
Moreira, Thaise S.G.; Creed, Joel C. (2012) Invasive, non-indigenous corals in a tropical rocky shore environment: No evidence for generalist predation, Journal of Experimental Marine Biology and Ecology 438: 7-13
Paz-Garcia, D. A.; Reyes-Bonilla, H.; Gonzalez-Peralta, A.; Sanchez-Alcantara, I. (2007) Larval release from Tubastraea coccinea in the Gulf of California, Mexico, Coral Reefs 26: 433
Pires-Teixeira, Larissa M. Neres-Lima, Vinicius Creed, Joel C. (2021) How do biological and functional diversity change in invaded tropical marine rocky reef communities?, Diversity 13(353): Published online
https://doi.org/ 10.3390/d13080353
Pires-Teixeira, Larissa M. Neres-Lima, Vinicius Creed, Joel C. (2021) How do biological and functional diversity change in invaded tropical marine rocky reef communities?, Diversity 18(353): Published online
https://www.mdpi.com/1424-2818/13/8/353
Rocha, Rosana M.; Cangussu, Leonardo C.; Braga, Mariana P. (2010) Stationary substrates facilitate bioinvasion in Paranaguá bay in southern Brazil, Brazilian Journal of Oceanography 58(Special Issue 4): 23-28
Sampaio, Cláudio L. S.; Miranda, Ricardo J.; Maia-Nogueira, Rodrigo; Nunes, José de Anchieta C.C. (2012) New occurrences of the nonindigenous orange cup corals Tubastraea coccinea and T. tagusensis (Scleractinia: Dendrophylliidae) in Southwestern Atlantic, Check List 8(3): 528-530
Silva, Rodrigo Vinagre, Catarina Kitahara, Marcelo V. Acorsi, Isabela V. Mizrahi, Damian Flores, Augusto A. V. (2019) Sun coral invasion of shallow rocky reefs: effects on mobile invertebrate assemblages in Southeastern Brazil, Biological Invasions 21(21): 1339–1350
Soares, Marcelo Oliveira; Xavier, Rafael de Lima, Francisco; Nalu Maia Dias, Monteiro da Silva, Maiara Queiroz; Pinto; de Lima Jadson; Xerez Barroso (2022) Alien hotspot: Benthic marine species introduced in the Brazilian semiarid coast, Marine Pollution Bulletin 174(113250): Published online
Soares, Marcelo, de Oliveira Salani, Sula Paiva, Sandra Vieira Andrade Braga, Marcus Davis (2020) Shipwrecks help invasive coral to expand range in the Atlantic Ocean, Marine Pollution Bulletin 158: Published online
https://doi.org/10.1016/j.marpolbul.2020.111394
Vançato, Yollanda Carolina da Silva Ferreira; Creed, Joel Christopher; Fleury, Beatriz Grosso (2023) Community structure of shallow tropical reefs undergoing invasion by Tubastraea spp. in a Brazilian Marine Protected Area, Aquatic Invasions 18(1): 39-57
https://doi.org/10.3391/ ai.2023.18.1.102938
Wells, John W. (1982) Notes on Indo-Pacific Scleractinian Corals. Part 9. 1 New Corals from the Galapagos Islands2, Pacific Science 36(2): 211-219