Invasion History
First Non-native North American Tidal Record: 1998First Non-native West Coast Tidal Record: 1998
First Non-native East/Gulf Coast Tidal Record:
General Invasion History:
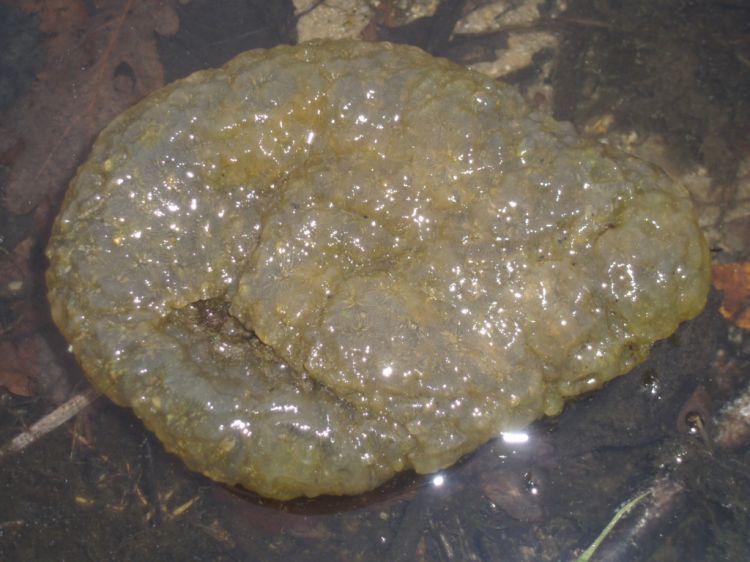
Pectinatella magnifica is a freshwater bryozoan described by Joseph Leidy in 1851 from the Delaware River in the vicinity of Philadelphia. It is widespread in Eastern North America from New Brunswick and Ontario (Ricciardi and Reiswig 1994) to Louisiana and Texas (Everitt 1975). Pectinatella magnifica forms large colonies in nutrient-rich habitats and is favored by the formation of reservoirs and ponds. It is often reported in the press as a mysterious 'blob'. As with other freshwater bryozoans, its statoblasts are tolerant of desiccation. It has been introduced to Europe, Asia and the Western US.
North American Invasion History:
Invasion History on the West Coast:
Pectinatella magnifica had previously been considered to range only as far west as Texas, but Marsh and Wood (2002) found it in 18 locations in Washington and Oregon, including waters in arid interior regions and wetter coastal and mountain localities. Its occurrence in the West, including the Columbia River watershed is apparently the result of human activities. This bryozoan was found in several Oregon tributaries in 1998, including the head of tide in the Willamette and Columbia Rivers (Bonneville Dam) (Marsh and Wood 2002), and was collected in the tidal river in the Columbia Slough near Portland in 2002 (Sytsma et al. 2004). Biologists and boaters along the tidal Columbia River have noticed increasing abundance of P. magnifica (US. Fish and Wildlife Service 2012). In 2005, this bryozoan was found in the Sacramento-San Joaquin River Delta, at the Oxbow Marina, by a US Fish and Wildlife Service Survey. It is now abundant in low-salinity habits in the San Francisco estuary (Jimenez et al. 2018).
Invasion History Elsewhere in the World:
Pectinatella magnifica has been introduced to European fresh waters, including France, Germany, and the Netherlands, possibly by shipping, fish introductions, or the ornamental water plants trade (Nehring 2002). It has also recently been introduced to Japan and Korea (Seo 1998).
Description
Pectinatella magnifica is a freshwater bryozoan which forms large, slimy, gelatinous, and transparent-to- brownish purple colonies up to 30-50 cm in diameter. Its zooids grow in rosette-like patches over a common gelatinous base. The lophophore bears 50-84 tentacles, and has conspicuous red pigment around the mouth. Its statoblasts are roughly circular, with a single row of flattened, hooked spines around the periphery (description from: Rogick 1935; Ricciardi and Reiswig 1994).
Taxonomy
Taxonomic Tree
| Kingdom: | Animalia | |
| Phylum: | Bryozoa | |
| Class: | Phylactolaemata | |
| Order: | Plumatellida | |
| Family: | Pectinatellidae | |
| Genus: | Pectinatella | |
| Species: | magnifica |
Synonyms
Potentially Misidentified Species
Ecology
General:
Life History- Pectinatella magnifica is a globular, soft-bodied, freshwater bryozoan composed of many individual zooids. Living colonies have a bubbly surface texture and rosettes of zooids can clearly be seen. The zooids feed by extending the ciliated tentacles of the lophophore as a funnel, creating a current, and driving food particles into their mouths. The food is guided down the tentacles and through the pharynx by the cilia. Larger food particles can be moved or captured by flicking or contracting the tentacles. The zooids are hermaphroditic, and produce large eggs, which are released into an embryo sac in the body cavity. This develops into a ciliated cystid sac, which buds off several zooids. The sac is a small ciliated colony which swims for a short period (less than 1-2 days). The cystid sac settles and the ciliated outer wall degenerates. The new colony continues to grow, but the parent zooids die, so that only the tips of the colony contain living zooids. Colonies also produce asexual propagules, called statoblasts, masses of cells enclosed by a chitinous shell. Statoblasts remain dormant and are resistant to desiccation and freezing. They have great potential for dispersal (Barnes 1983). Colonies of Pectinatella magnifica die off in winter. Dying or dead colonies in autumn become smooth, slimy and transparent masses of gelatin, peppered with black statoblasts. (Ricciardi and Reiswig 1994; Judy Winston, personal communication).
Ecology- Pectinatella magnifica inhabits lakes and rivers, at 6.8-9.4 pH and 4-32 °C (Everitt 1975; Ricciardi and Reiswig 1994). It has been found attached to rocks, wood, aquatic vegetation, sponges, freshwater mussels, and other bryozoans (Ricciardi and Reiswig 1994).
Food:
Phytoplankton
Consumers:
caddisflies, crayfish, fishes
Trophic Status:
Suspension Feeder
SusFedHabitats
| General Habitat | Nontidal Freshwater | None |
| General Habitat | Fresh (nontidal) Marsh | None |
| General Habitat | Coarse Woody Debris | None |
| General Habitat | Tidal Fresh Marsh | None |
| General Habitat | Canals | None |
| Salinity Range | Limnetic | 0-0.5 PSU |
| Salinity Range | Oligohaline | 0.5-5 PSU |
| Tidal Range | Subtidal | None |
| Vertical Habitat | Epibenthic | None |
Tolerances and Life History Parameters
| Minimum Temperature (ºC) | 0 | Field- Based on occurrence in Canadian lakes.Large colonies break up at 10-12 C, but smaller colonies in Quebec tolerate 9 C. Statoblasts tolerate temberatures below freezing (Ricciardi and Reiswig 1994). |
| Maximum Temperature (ºC) | 32 | Field, Louisiana, Everitt 1975 |
| Minimum Salinity (‰) | 0 | This is a freshwater species. |
| Maximum Salinity (‰) | 0.4 | Field, Louisiana, Everitt 1975 |
| Minimum pH | 6.8 | Field, Ricciardi and Reiswig 1994 |
| Maximum pH | 9.4 | Field, Ricciardi and Reiswig 1994 |
| Minimum Reproductive Temperature | 9 | Field, in Canadian lakes (Ricciardi and Reiswig 1994) |
| Maximum Length (mm) | 1,300 | 'I've seen colonies at least 4 feet high attached to boathouse pilings in a Virginia lake in autumn.' (Judy Winston, personal communication) |
| Minimum Height (mm) | 60 | Colony height, Wood 1989 |
| Broad Temperature Range | None | Cold temperate-Warm temperate |
| Broad Salinity Range | None | Nontidal Limnetic-Tidal Limnetic |
General Impacts
Freshwater bryozoans, such as Pectinatella magnifica, sometimes become so abundant that they clog fishing nets, foul power plant water systems, and obstruct municipal water systems (Ricciardi and Reiswig 1994). However, impacts have not been reported from the Columbia River (Systma et al. 2004).Parasite/Predator Vector: Pectinatella magnifica is one of many freshwater bryozoans which may serve as an intermediate host of Tetracapsula bryosalmonae, a myxozoan parasite which causes Proliferative Kidney Disease in salmonids. However, it was found in only one of the surveyed hatcheries (Folsom Lake, California) which had an outbreak of the disease. Other bryozoans may be more frequent hosts of this parasite (Okamura and Wood 2002).
Regional Distribution Map
Non-native
Native
Cryptogenic
Failed
| Bioregion | Region Name | Year | Invasion Status | Population Status |
|---|---|---|---|---|
| P260 | Columbia River | 1998 | Non-native | Established |
| GL-I | Lakes Huron, Superior and Michigan | 0 | Native | Established |
| GL-II | Lake Erie | 0 | Native | Established |
| GL-III | Lake Ontario | 0 | Native | Established |
| M090 | Delaware Bay | 0 | Native | Established |
| M130 | Chesapeake Bay | 0 | Native | Established |
| L095 | _CDA_L095 (Cedar-Portage) | 0 | Native | Established |
| L014 | _CDA_L014 (Beartrap-Nemadji) | 0 | Native | Established |
| P090 | San Francisco Bay | 2005 | Non-native | Established |
| NA-S3 | None | 0 | Native | Established |
| NEP-IV | Puget Sound to Northern California | 1998 | Non-native | Established |
| NA-ET3 | Cape Cod to Cape Hatteras | 0 | Native | Established |
| NEP-V | Northern California to Mid Channel Islands | 2005 | Non-native | Established |
Occurrence Map
| OCC_ID | Author | Year | Date | Locality | Status | Latitude | Longitude |
|---|---|---|---|---|---|---|---|
| 767939 | Ruiz et al., 2015 | 2011 | 2011-09-26 | Pittsburg Marina, San Francisco Bay, CA, California, USA | Non-native | 38.0346 | -121.8829 |
| 768257 | Ruiz et al., 2015 | 2012 | 2012-09-10 | Sacramento Marina, San Francisco Bay, CA, California, USA | Non-native | 38.5648 | -121.5184 |
| 768260 | Ruiz et al., 2015 | 2012 | 2012-09-13 | Port of Stockton, San Francisco Bay, CA, California, USA | Non-native | 37.9589 | -121.3609 |
References
Barnes, Robert D. (1983) Invertebrate Zoology, Saunders, Philadelphia. Pp. 883Cheng, Brian S.;; Blumenthal, Jeffrey; Chang, Andrew L.; Barley, Jordanna; Matthew C. Ferner, K; Nielsen, arina J.;Ruiz, Gregory M.; Zabin, Chela J. (2021) Severe introduced predator impacts despite attempted functional eradication, Biological Invasions Published online: <missing location>
Everitt, Betty (1975) Fresh-water Ectoprocta: distribution and ecology of five species in southeastern Louisiana, Transactions of the American Microscopical Society 94(1): 130-134
Grigorovich, Igor A; Korniushin, Alexei V.; Gray, Derek K.; Duggan, Ian C.; Colautti, Robert I.; MacIsaac, Hugh J. (2003) Lake Superior: an invasion coldspot?, Hydrobiologia 499: 191-210
Grottoli, Andrea G. and 11 authors (2018) Coral physiology and microbiome dynamics under combined warming and ocean acidification, None 13: Published online
https://doi.org/10.1371/journal.pone.0191156
Hamelin, Kayla M.; Castaneda, Rowshyra A.; Ricciardi, Anthony (2016) Cryptic invaders: nonindigenous and cryptogenic freshwater Bryozoa and Entoprocta in the St. Lawrence River, Biological Invasions Published online: <missing location>
Jimenez, H.; Keppel, E.; Chang, A. L.; Ruiz, G. M. (2018) Invasions in marine communities: Contrasting species richness and community composition across habitats and salinity, Estuarine and Coasts 41: 484-494
Liu, Wenliang; Liang, Xiaoli ; Zhu, Xiaojing (2015) A new record and mitochondrial identification of Synidotea laticauda Benedict, 1897 (Crustacea: Isopoda: Valvifera: Idoteidae) from the Yangtze Estuary, China, Zootaxa 4294: 371-380
Marsh, Terrence; Wood, Timothy S. (2002) Bryozoan Studies 2001, Swets & Zeitlinger, Lisse. Pp. <missing location>
Nehring, Stefan (2002) Invasive aquatic species of Europe: Distribution, impacts and management, Kluwer Academic Publishers, Dordrecht, Boston, London.. Pp. 373-383
Okamura, B.; Wood, T. S. (2002) Bryozoans as hosts for Tetracapsula bryosalmonae, the PKX organism, Journal of Fish Diseases 25: 469-475
Ricciardi, Anthony; Reiswig, Henry M. (1994) Taxonomy, distribution, and ecology of the freshwater bryozoans (Ectoprocta) of eastern Canada, Canadian Journal of Zoology 72: 339-359
Rogick, Mary D. (1935) Studies on Freshwater Bryozoa: II. The Bryozoa of Lake Erie., Transactions of the American Microscopical Society 54(3): 245-263
Salmon, Terry and 21 authors 2014-2022 California Fish Website. https://calfish.ucdavis.edu/
Seo, Ji Eun (1998) Taxonomy of the marine bryozoans from Namheado Island and its adjacent waters, Korea., Korean Journal of Systematic Zoology 14(4): 415-424
Sytsma, Mark D.; Cordell, Jeffrey R.; Chapman, John W.; Draheim, Robyn, C. (2004) <missing title>, Center for Lakes and Reservoirs, Portland State University, Portland OR. Pp. <missing location>
U.S. National Museum of Natural History 2002-2021 Invertebrate Zoology Collections Database. http://collections.nmnh.si.edu/search/iz/
USGS Nonindigenous Aquatic Species Program 2003-2024 Nonindigenous Aquatic Species Database. https://nas.er.usgs.gov/
Wood, T. S. (2002) Bryozoan Studies 2001, Swets & Zeiltinger, Lisse, Netherlands. Pp. 339-345
Wood, Timothy S. (1989) Ectoproct Bryozoans of Ohio, Bulletin of the Ohio Biological Survey 8(2): 1-67
Wood, Timothy S. (1991) Ecology and Classification of North American Freshwater Invertebrates, Academic Press, San Diego. Pp. 481-499