Invasion History
First Non-native North American Tidal Record: 2002First Non-native West Coast Tidal Record:
First Non-native East/Gulf Coast Tidal Record: 2002
General Invasion History:
Celleporaria pilaefera is an encrusting bryozoan with a broad range in the Indo-Pacific, including Mauritius, the Red Sea, India, the Philippines (type locality), and Vanuatu (Swami and Karande 1994), although whether this distribution represents its native range is not known.
North American Invasion History:
Invasion History on the East Coast:
In 2002, Celleporaria pilaefera was found growing in low abundance (on two plates, one colony each) at Mayport Naval Station, Jacksonville, Florida, on the St. Johns River estuary (McCann et al. 2007). It has not yet been found in other East Coast locations.
Invasion History in Hawaii:
In 1997 , C. pilaefera was collected in Honolulu Harbor and Keehi Lagoon, Oahu (Coles et al. 1999a). It was also found in Kaneohe Bay (Coles et al. 2002a) and Waikiki (Coles et al. 2002b). It is considered introduced in the Hawaiian Islands, probably by ship hull fouling (James T. Carlton, personal communication). A population was also found at the remote Johnston Atoll, 1325 km southwest of Honolulu (Coles et al. 2001).
Invasion History Elsewhere in the World:
In 1977, Celleporaria pilaefera was collected on oyster aquaculture cages, in Malta, in the Mediterranean Sea (Agius et al. 1977). There are no further records of this bryozoan in Malta or elsewhere in the Mediterranean and oyster aquaculture has ceased (Sciberras and Schembri 2007). In 2007, it was found on the hull of a wrecked fishing vessel off the coast of New Zealand (Williams et al. 2008). Aside from the records in Jacksonville, Florida, Hawaii, and Johnston Atoll there are no known established populations of this bryozoan.
Description
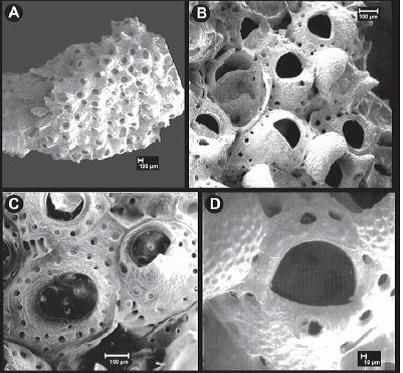
Celleporaria pilaefera forms encrusting colonies. Its zooids are irregularly polygonal in outline, and about 0.40 mm long by 0.35 mm wide. The polypide in each zooid has 13-16 brown-tipped tentacles. The frontal wall is convex, with a raised unperforated, granular, calcified region centered around the orifice, and large round pores scattered at or near the zooid margins. The orifice is large relative to zooid size, and slightly more than semi-circular distally, with a smooth and shallowly concave proximal margin, with small condyles at the hinge. Below the orifice is a mound (umbo) which forms the base of an avicularium. This bryozoan does not have avicularia at the boundaries between zooids, but does have occasional scattered avicularia. The ovicells are cap-shaped, and open with unperforated granular calcification. In some cases, ovicells are asymmetric over the operculum (Description from McCann et al. 2007).
Taxonomy
Taxonomic Tree
| Kingdom: | Animalia | |
| Phylum: | Bryozoa | |
| Class: | Gymnolaemata | |
| Order: | Cheilostomata | |
| Suborder: | Ascophora | |
| Family: | Celleporariidae | |
| Genus: | Celleporaria | |
| Species: | pilaefera |
Synonyms
Potentially Misidentified Species
Ecology
General:
Life History- Celleporaria pilaefera is an encrusting, calcified bryozoan, composed of many individual zooids. The zooids feed by extending the ciliated tentacles of the lophophore as a funnel, creating a current, and driving food particles into their mouths. The food is guided along the tentacles and through the pharynx by the cilia. Larger food particles can be moved or captured by flicking or contracting the tentacles (Barnes 1983). Larvae of other species of Celleporaria are non-feeding and have a short period of planktonic development (Hall 1982; Santagata 2008). Larvae settle on a substrate and metamorphose into the first zooid of a colony, an ancestrula (Barnes 1983).
Ecology- Celleporaria pilaefera is known from coral reefs, oyster aquaculture cages, fouling plates, and ships' hulls (Agius et al. 1977; Winston 1986; McCann et al. 2007; Williams et al. 2008). In the St. Johns River estuary, Florida, it was collected at a salinity of 30 PSU (McCann et al. 2007), and its known range suggests a preference for marine salinities and a tropical-subtropical climate.
Food:
Phytoplankton, detritus
Trophic Status:
Suspension Feeder
SusFedHabitats
| General Habitat | Marinas & Docks | None |
| General Habitat | Coral reef | None |
| General Habitat | Rocky | None |
| Salinity Range | Polyhaline | 18-30 PSU |
| Salinity Range | Euhaline | 30-40 PSU |
| Tidal Range | Subtidal | None |
| Vertical Habitat | Epibenthic | None |
Tolerances and Life History Parameters
| Broad Temperature Range | None | Warm Temperate-Tropical |
| Broad Salinity Range | None | Polyhaline-Euhaline |
General Impacts
Impacts of introduced populations of Celleporaria pilaefera have not been studied.Regional Impacts
| MED-IV | None | Economic Impact | Fisheries | ||
| Fouling aquaculture cages | |||||
Regional Distribution Map
Non-native
Native
Cryptogenic
Failed
Occurrence Map






References
Agius, C.; Schembri, P. J.; Jaccarini, V. (1977) A preliminary report on organsims fouling oyster cultures in Malta., Memorie di Biologia Marina e di Oceanographia 7(3-4): 51-59Barnes, Robert D. (1983) Invertebrate Zoology, Saunders, Philadelphia. Pp. 883
Carlton, James T.; Eldredge, Lucius G. (2015) Update and revisions of the marine bioinvasions of Hawai‘i: The introduced and cryptogenic marine and estuarine animals and plants of the Hawaiian archipelago, Bishop Museum Bulletin in Cultural and Environmental Studies 9: 25-47
Coles S. L., DeFelice R. C., Eldredge, L. G. (1999a) Nonindigenous marine species introductions in the harbors of the south and west shores of Oahu, Hawaii., Bishop Museum Technical Report 15: 1-212
Coles S. L., DeFelice R. C., Eldredge, L. G. (2002b) Nonindigenous marine species at Waikîkî and Hawai`i kai, Oahu, Hawai`i, Bishop Museum Technical Report 25: 1-255
Coles, S. L.; DeFelice, R. C. : Eldredge, L. G. (2002a) Nonindigenous marine species in Kaneohe Bay, Oahu, Hawai`i, Bishop Museum Technical Report 24: 1-364
Coles, S. L.; DeFelice, R. C.; Minton, D. (2001) Marine species survey of Johnston Atoll, Central Pacific Ocean, June 2000, Bishop Museum Technical Report 19: 1-59
Cranfield, H.J.; Gordon, D.P.; Willan, R.C.; Marshall, B.A; Battershill, C.N.; Francis, M.P.; Nelson, W.A.; Glasby, C.J.; Read, G.B. (1998) <missing title>, The National Institute of Water and Atmospheric Research, New Zealand. Pp. <missing location>
Gordon, Dennis P. (2016) Bryozoa of the South China Sea: an overview, Raffles Bulletin of Zoology 34: 604-618
Gordon, Dennis P.; Hosie, Andrew M.; Carter, Michelle C. (2008) Post-2000 detection of warm-water alien bryozoan species in New Zealand- The significance of recreational vessels, Virginia Museum of Natural History Special Publication 15: 37-48
Hall, D.N. (1982) Larval release in Celleporaria apiculata (Busk) (Bryozoa: Ascophora), Journal of Natural History 16(2): 195-200
McCann, Linda D.; Hitchcock, Natasha Gray; Winston, Judith E.; Ruiz, Gregory M. (2007) Non-native bryozoans in coastal embayments of the southern United States: new records for the western Atlantic., Bulletin of Marine Science 80(2): 319-342
Ruiz, Gregory M.; Geller, Jonathan (2018) Spatial and temporal analysis of marine invasions in California, Part II: Humboldt Bay, Marina del Re, Port Hueneme, and San Francisco Bay, Smithsonian Environmental Research Center & Moss Landing Laboratories, Edgewater MD, Moss Landing CA. Pp. <missing location>
Santagata, Scott (2008) The morphology and evolutionary significance of the ciliary fields and musculature among marine bryozoan larvae, Journal of Morphology 269: 349-364
Sciberras, M.; Schembri, P. J. (2007) A critical review of records of alien marine species from the Maltese Islands and surrounding waters (Central Mediterranean)., Mediterranean Marine Science 8(1): 41-66
Swami, B. S.; Karande, A. A. (1994) Encrusting bryozoans in Karwar waters, central west coast of India, Indian Journal of Marine Sciences 23: 170-172
Virgili. Riccardo; Tandua, Valentina; Katsanevakis, Stelos; Terlizzi, Francesco; Villani, Guido; Fontano. Angelo; Crocetta, Fabio (2022) The Miseno Lake (Central-Western. Mediterranean Sea): An overlooked reservoir of non-indigenous and cryptogenic ascidians in a marine reserve, Frontiers in Marine Science 9(866906): Published online
Williams, Rissa; Gould, Brendan; Christian, Sheree (2008) Shipwrecks: an international biosecurity risk?, Surveillance 35(1): 1-6
Winston, Judith E. (1986) An annotated checklist of coral-associated bryozoans, American Museum Novitates 2859: 1-39